IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii
What is the formula to calculate the radius of an orbit of the atom and velocity of the specific shell of the atom.

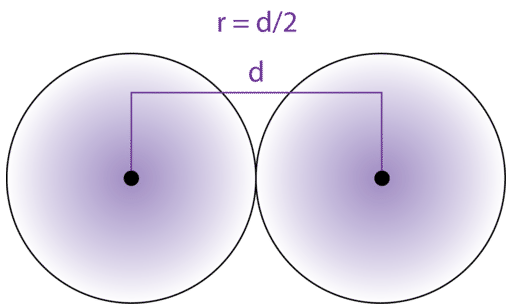
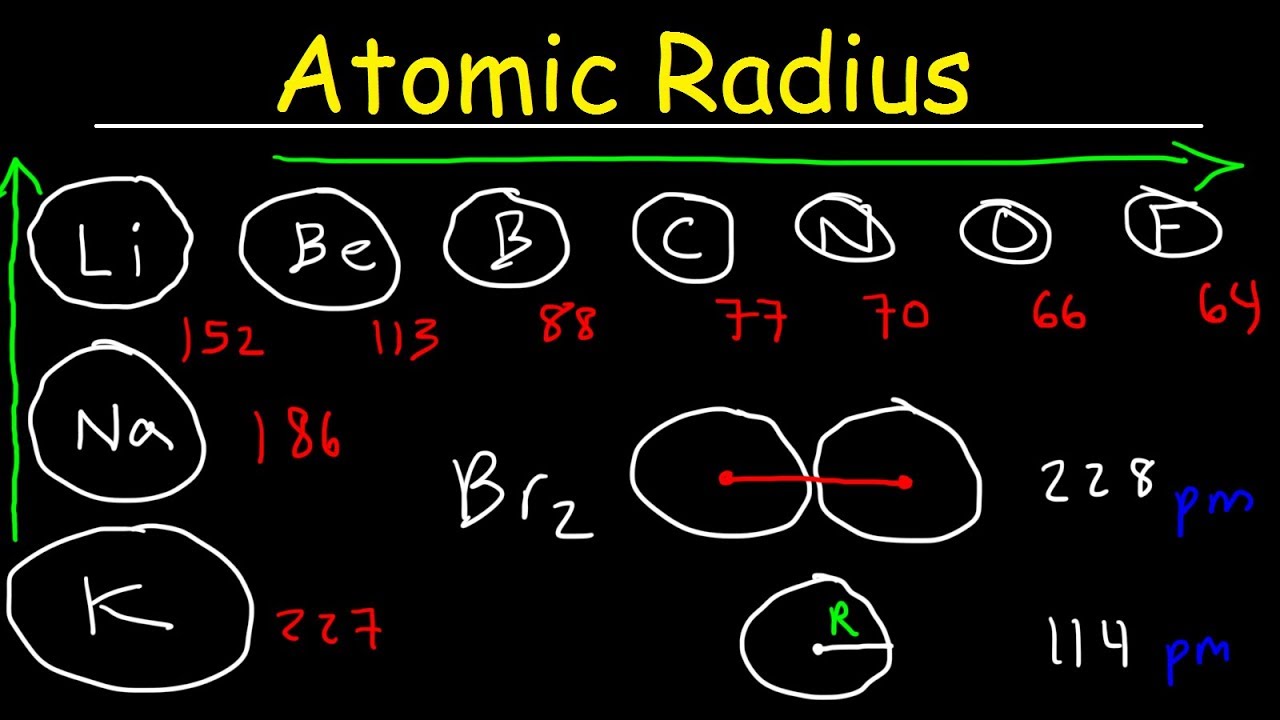
Atomic radius of `Li is 1.23 Å` and ionic radius of `Li^(+)` is `0.76 Å`. Calculate the percentage - YouTube

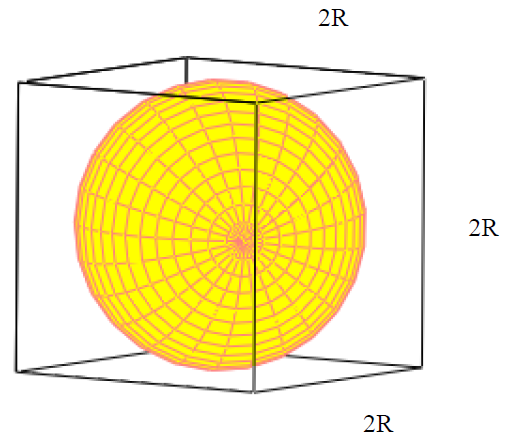
Calculate the atomic radius of elementary silver which crystallises in face centred cubic lattice with unit cell edge length 4.086 xx 10^(-10) m.

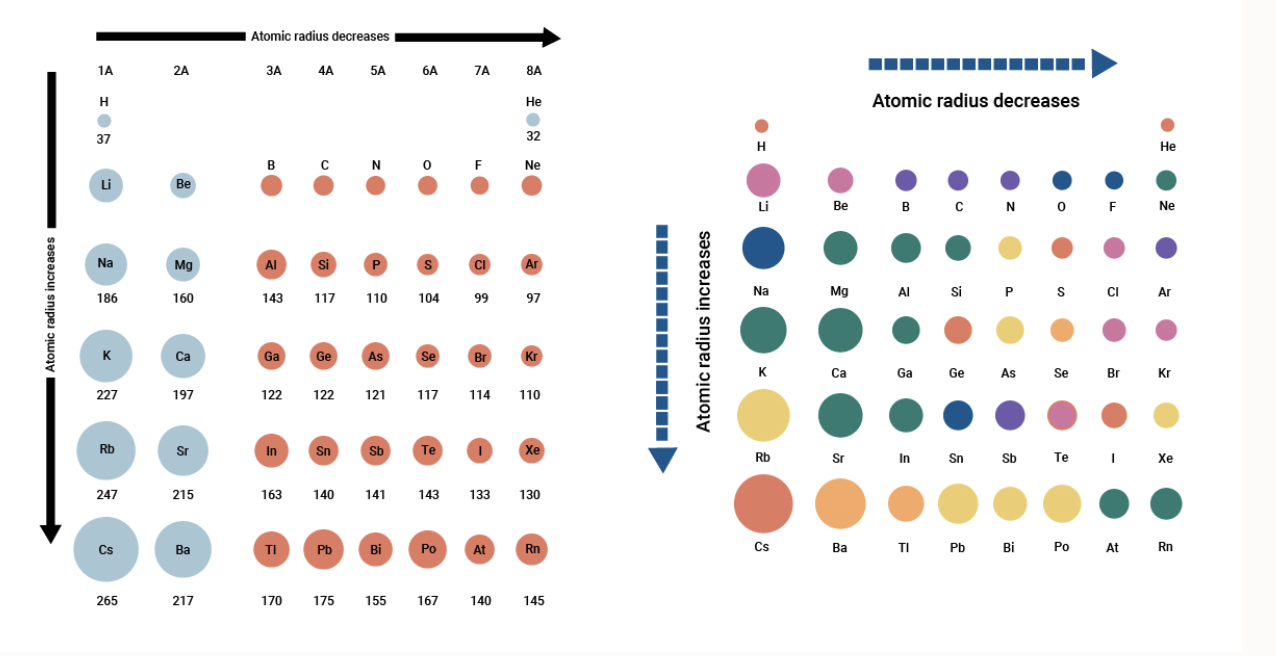
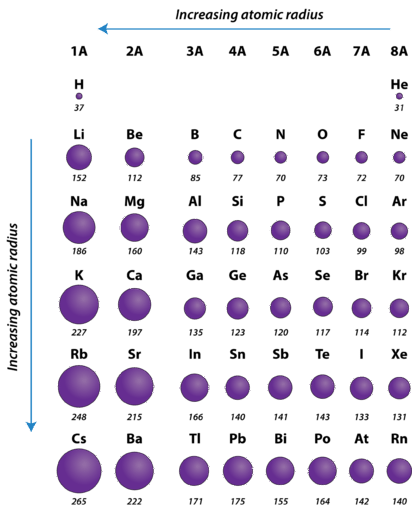
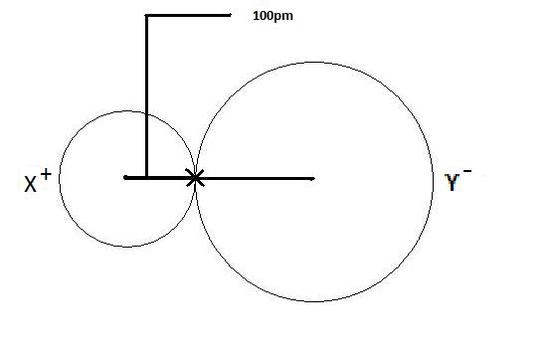
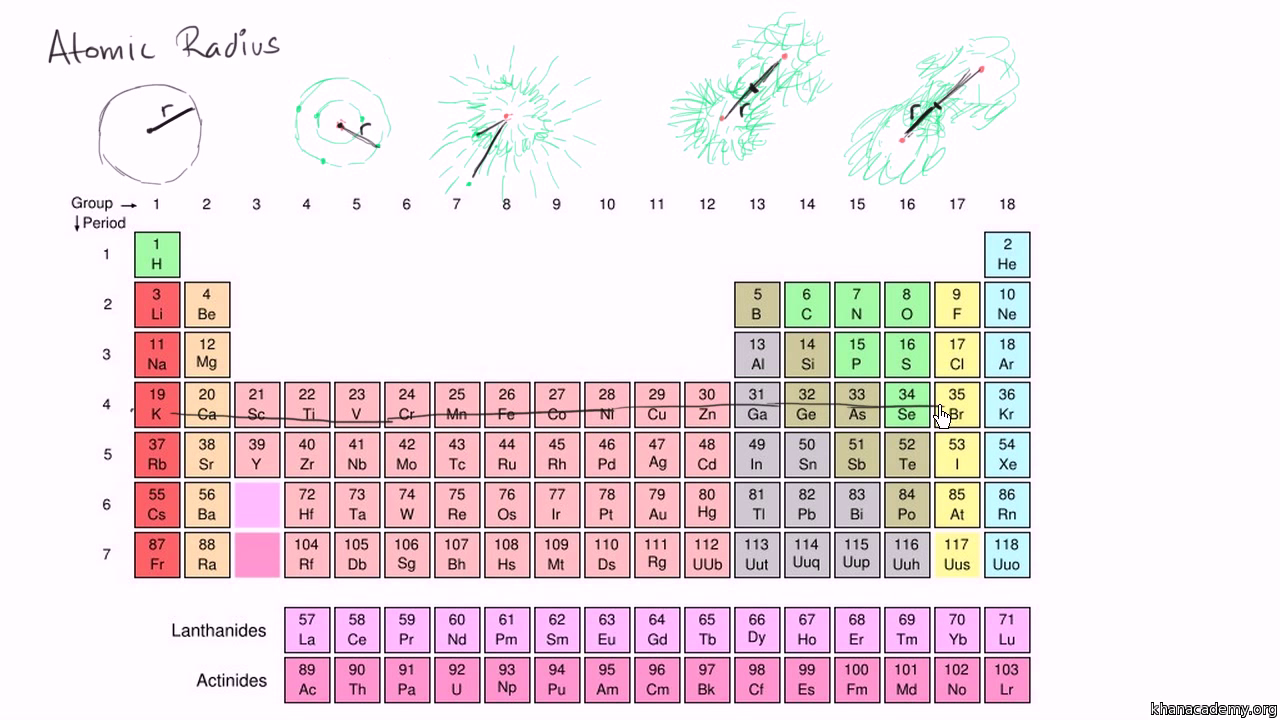
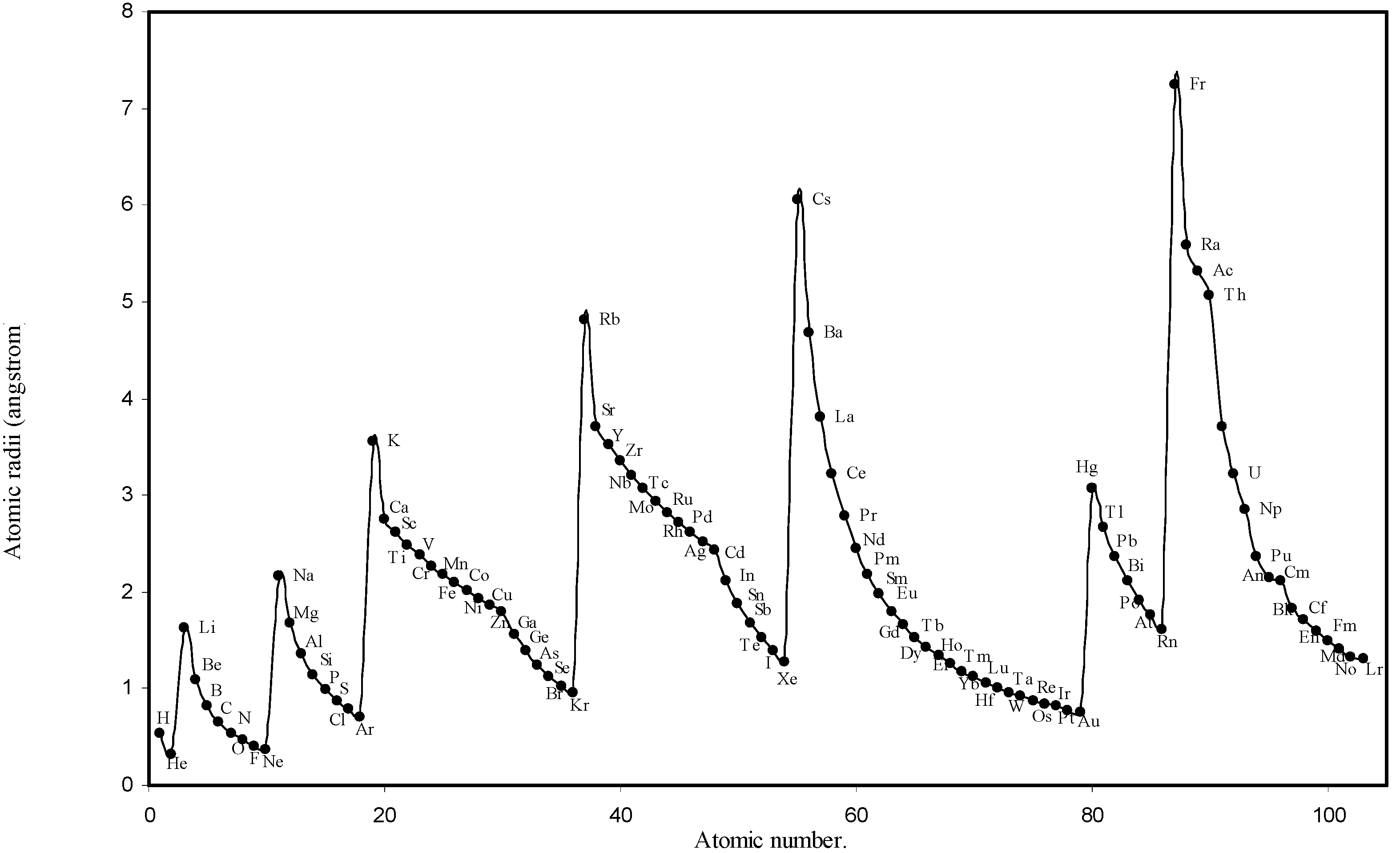
How to arrange the following atoms and ions in order of increasing atomic size?: "Rb", "Ag", "O"^(-2), "Al", "O", "Cs", "Al"^(+3), "Si" | Socratic

Calculate the radius of He atoms if its van der Waal's constant 'b' is 24mL `"mol"^(-1)`. (Note: mL= - YouTube