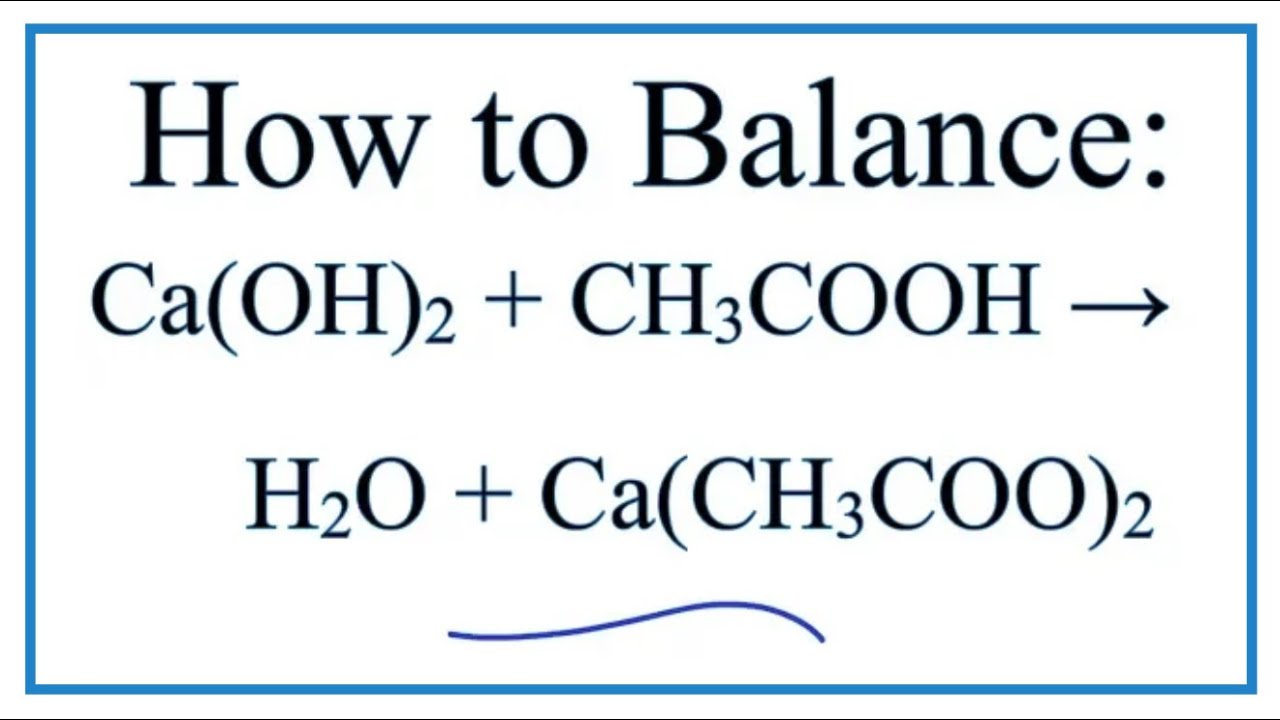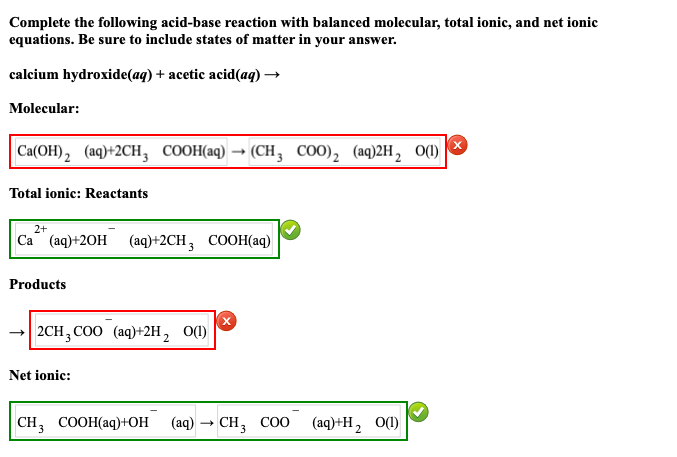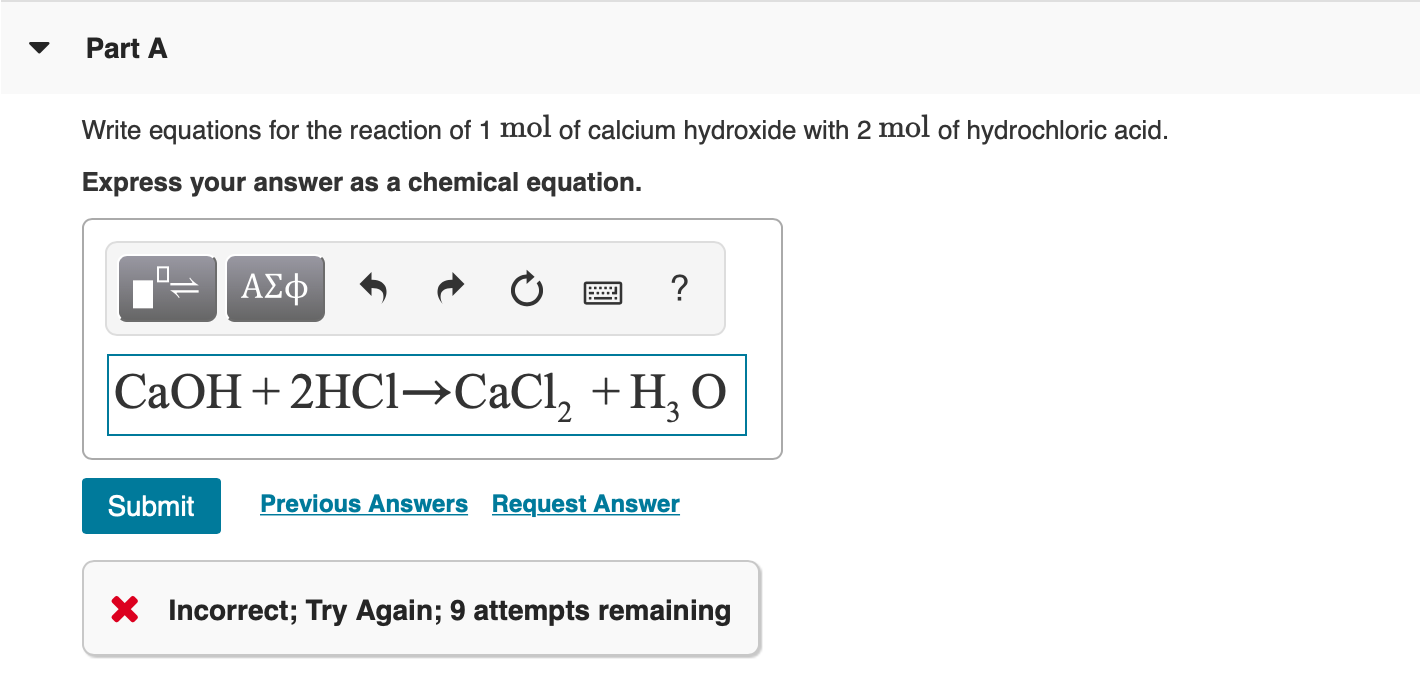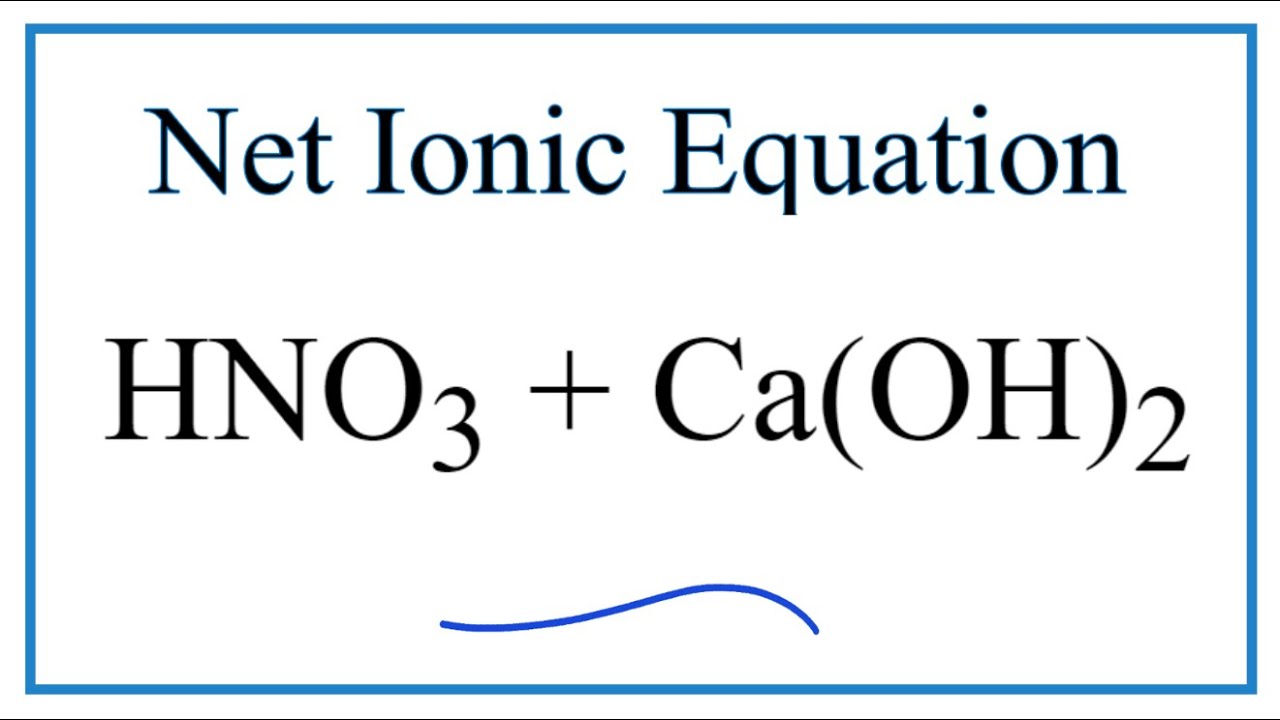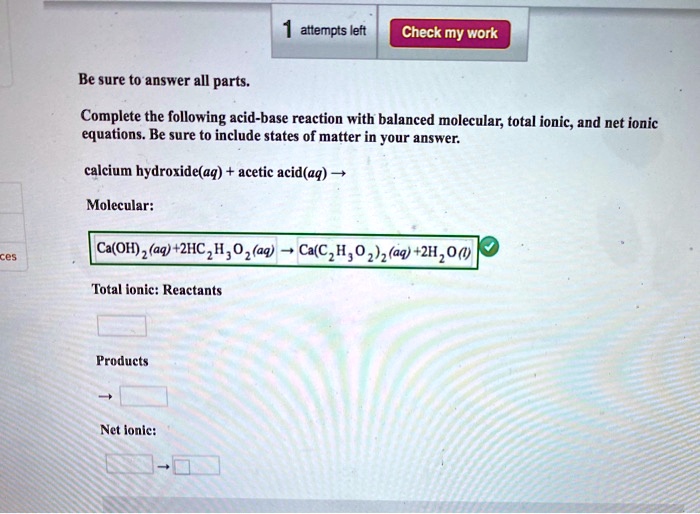
SOLVED: attempts left Check my work Be sure to answer all parts. Complete the following acid-base reaction with balanced molecular; total ionic, and net ionic equations. Be sure to include states of

ACIDS & BASES module i.An acid is a chemical substance that …………………in water to produce ………………. ions. ii.A base is a chemical substance that ………………in. - ppt download

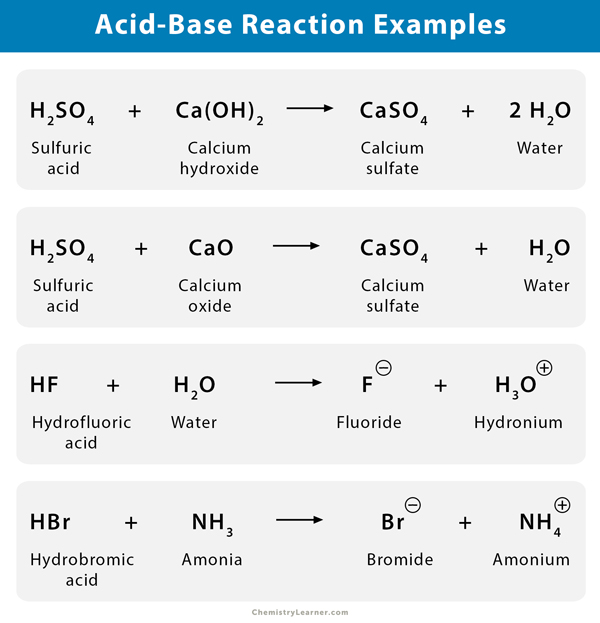
SOLVED: 9 . Write a balanced neutralization reaction between the following acids and bases a. Carbonic acid and Potassium hydroxide Phosphoric acid and Calcium hydroxide c. Nitric acid and Aluminum hydroxide d.