
SOLVED:Calculate the number of atoms in each element. a. 25.8 g of Hg b. 0.0340 g of Zn c. 150 g of Ar d. 0.124 g of Mg
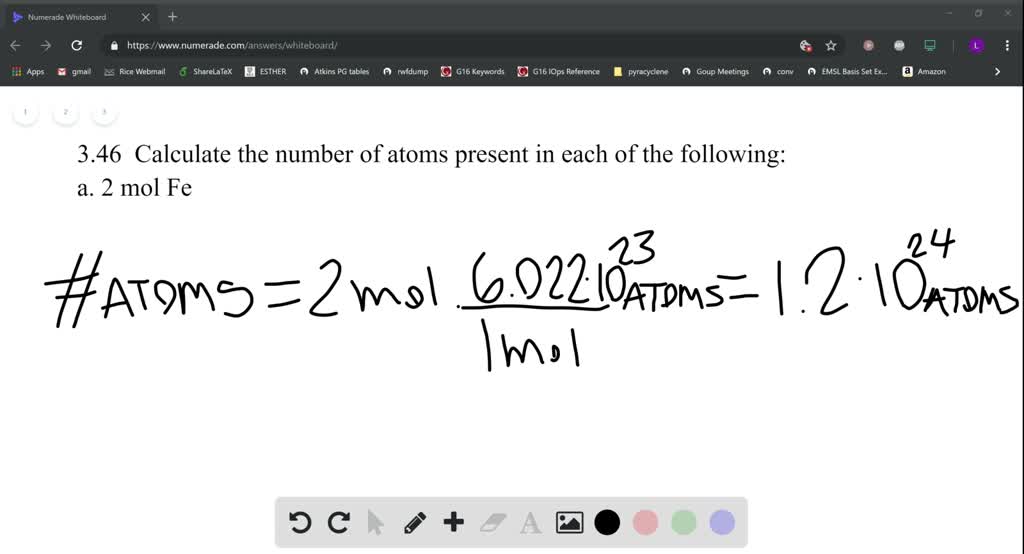
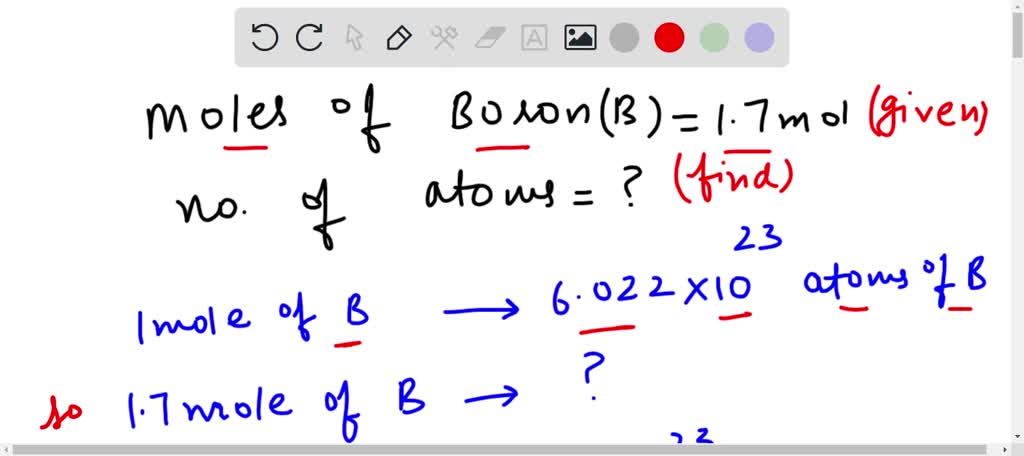
SOLVED:Calculate the number of atoms present in each of the following: a. 2 mol Fe b. 40.1 g Ca, which has an atomic mass of 40.08 amu c. 4.5 mol of boron-11

Calculate the number of atoms present in 2 gram of crystal which has face centred cubic (FCC) crystal lattice having edge length of 100 pm and density 10gcm^-3

Calculate the number of atoms of each element in 3.42 g of sucrose (C(12)H(22)O(11)) and the total number of atoms.

Calculate number of atoms in each of the following" i. 0.5 mol atom of nitorgen ii. 0.2 mol molecules of hydrogen iii. 3.2 g of sulphur Calculate number of molecules in each





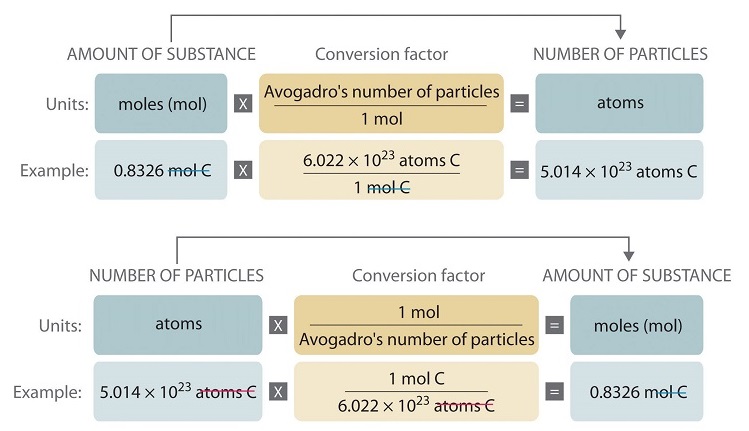
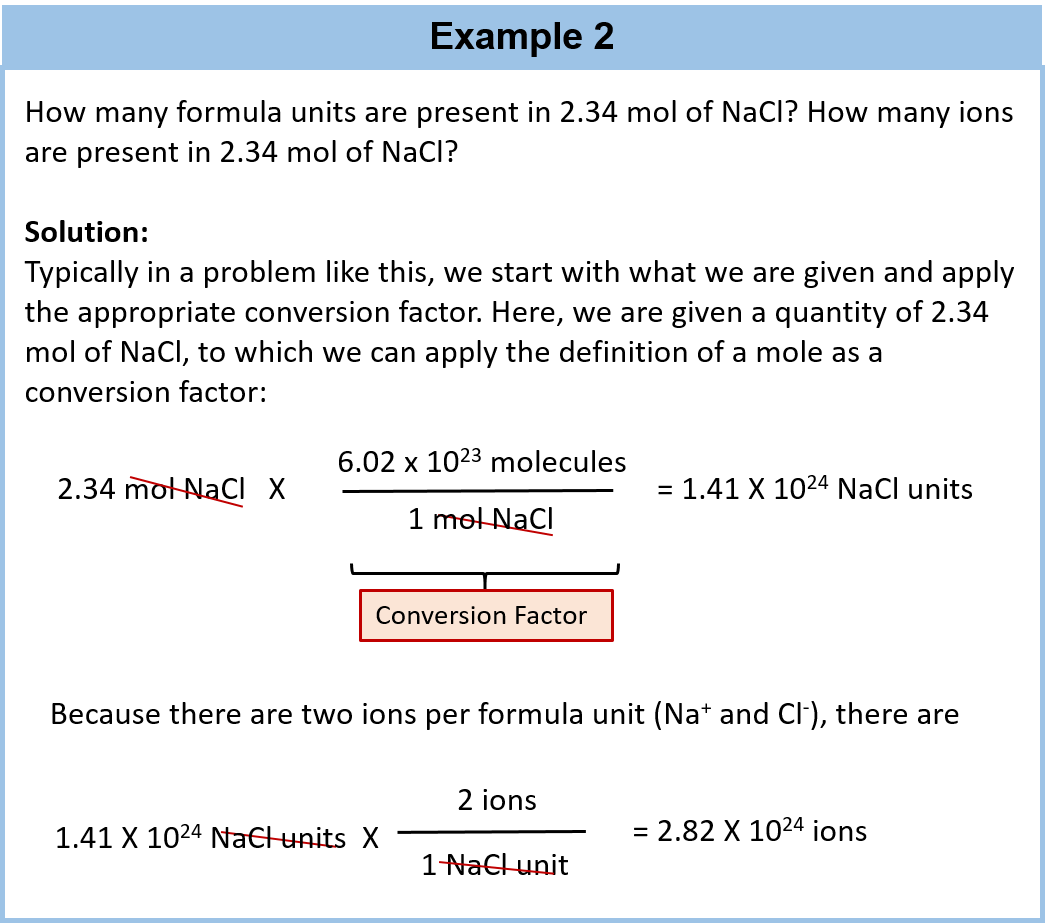







![Calculate no of atoms of each type in 1.8g glucose [Give ans in terms of NA ] Calculate no of atoms of each type in 1.8g glucose [Give ans in terms of NA ]](https://haygot.s3.amazonaws.com/questions/1294799_1324580_ans_2473354570594c17b649aed6ddb7fffd.jpg)