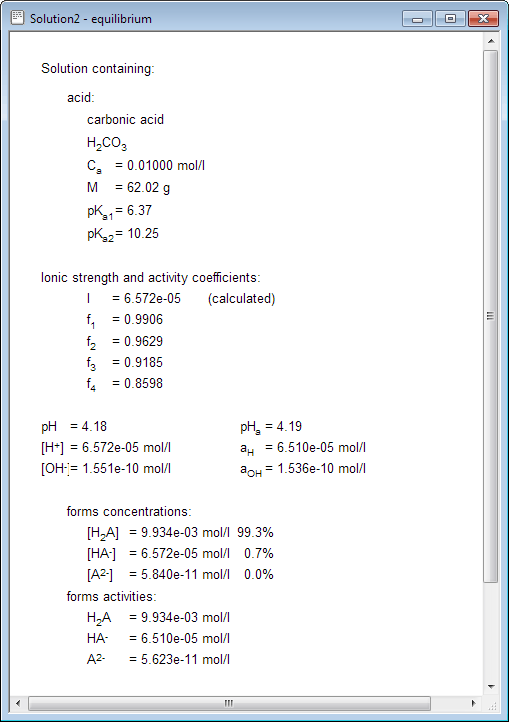
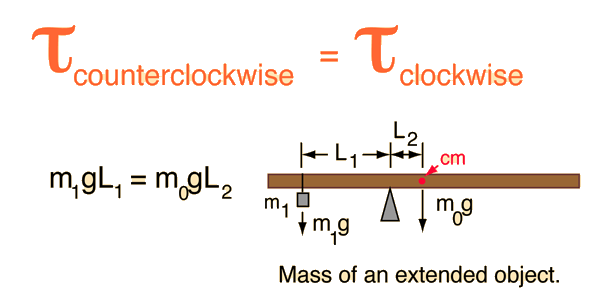
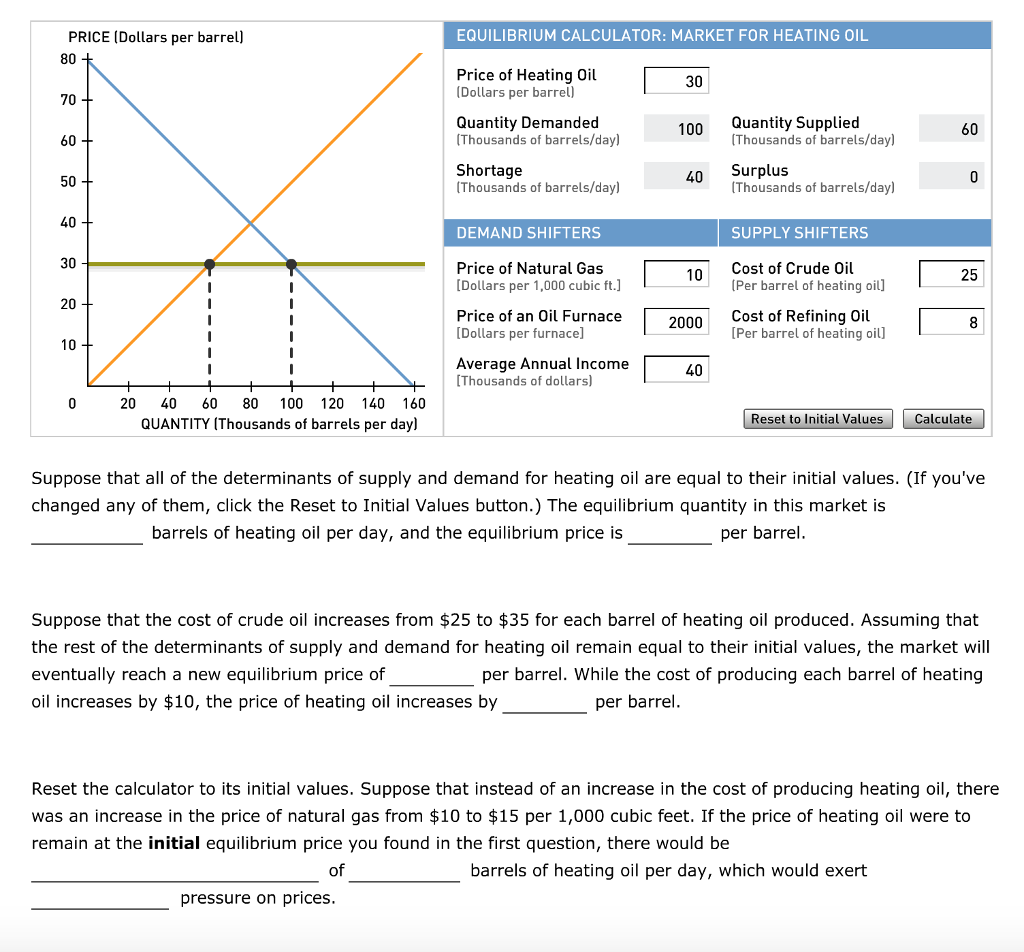
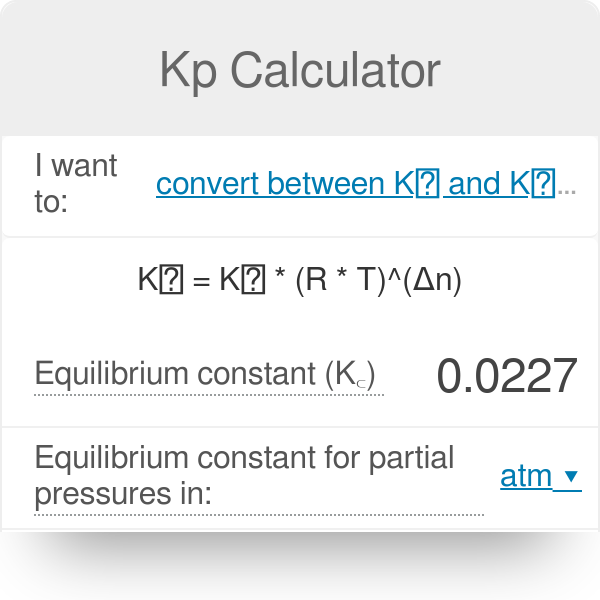
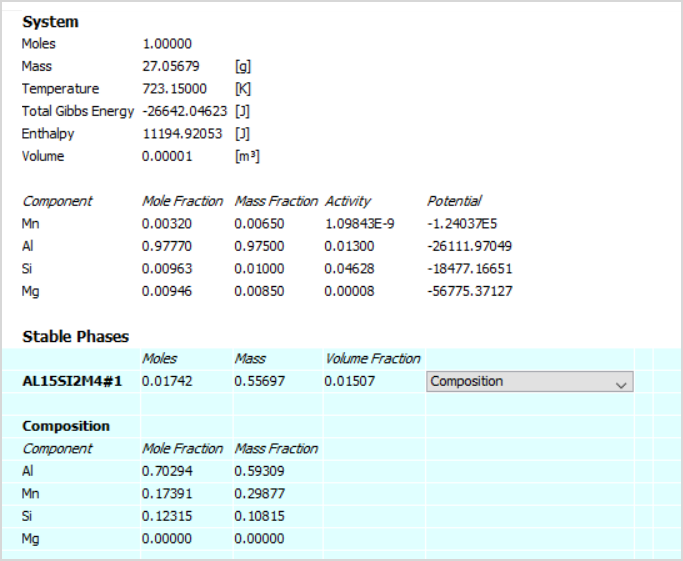
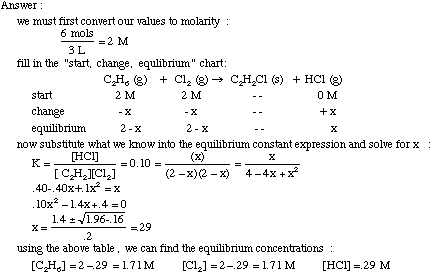A Simple Method To Calculate the Temperature Dependence of the Gibbs Energy and Chemical Equilibrium Constants | Journal of Chemical Education

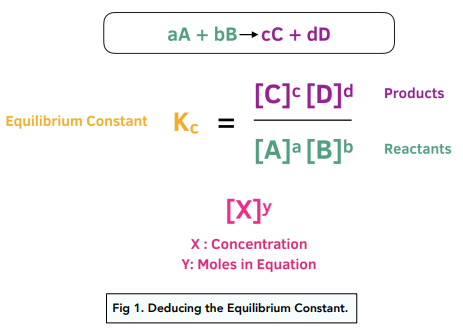
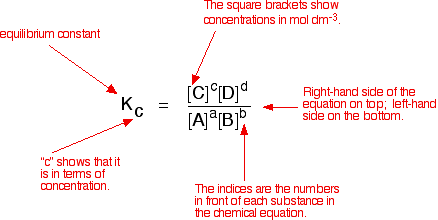
Calculate the equilibrium constant (Kc) for the formation of NH3 in the following reaction: N2 (g) + 3H2 (g) 2NH3 (g) At equilibrium, the concentration of NH3, H2 and N2 are 1.2 ×