
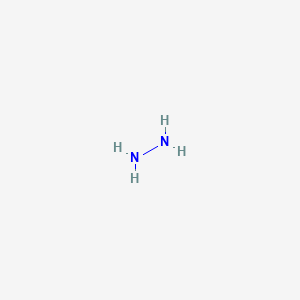
SOLVED: Hydrazine (N2H4) is a weak base. Write an equation for its dissociation in an aqueous solution: Label the acid, base conjugate acid, and conjugate base. (6 pts) What is the Kb
![PDF] Synthesis, crystal structure, and antidiabetic property of hydrazine functionalized Schiff base: 1,2-Di(benzylidene)hydrazine | Semantic Scholar PDF] Synthesis, crystal structure, and antidiabetic property of hydrazine functionalized Schiff base: 1,2-Di(benzylidene)hydrazine | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39bb0190c68df2ae004d05d1f71f4cce7460deed/2-Table1-1.png)
PDF] Synthesis, crystal structure, and antidiabetic property of hydrazine functionalized Schiff base: 1,2-Di(benzylidene)hydrazine | Semantic Scholar
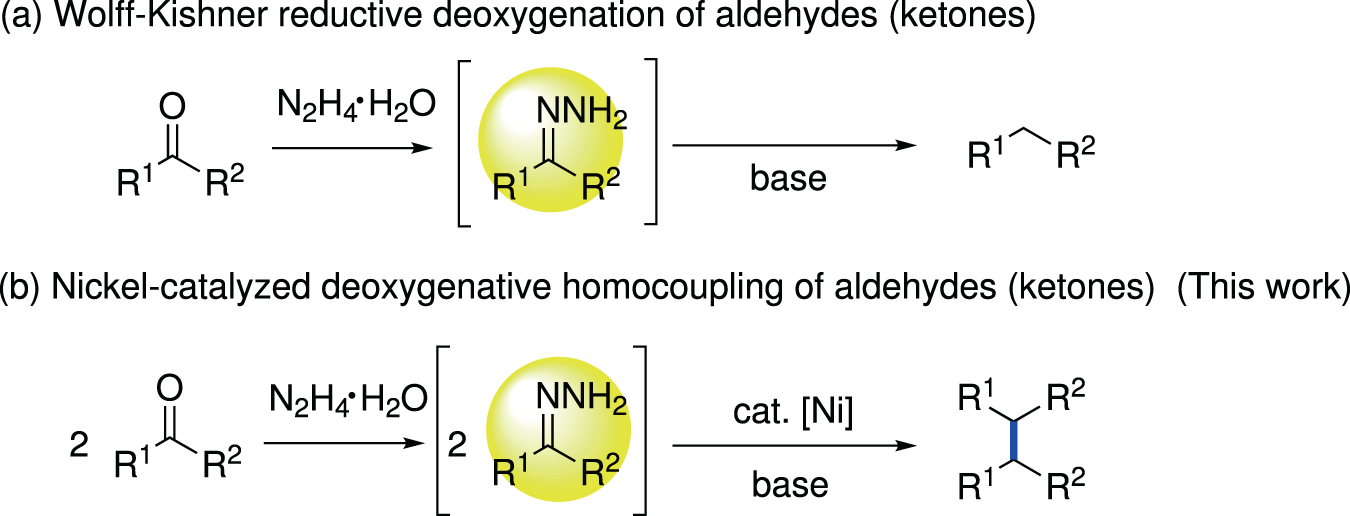
C(sp3)−C(sp3) bond formation via nickel-catalyzed deoxygenative homo-coupling of aldehydes/ketones mediated by hydrazine | Nature Communications

Electrochemical Strategy for Hydrazine Synthesis: Development and Overpotential Analysis of Methods for Oxidative N–N Coupling of an Ammonia Surrogate | Journal of the American Chemical Society
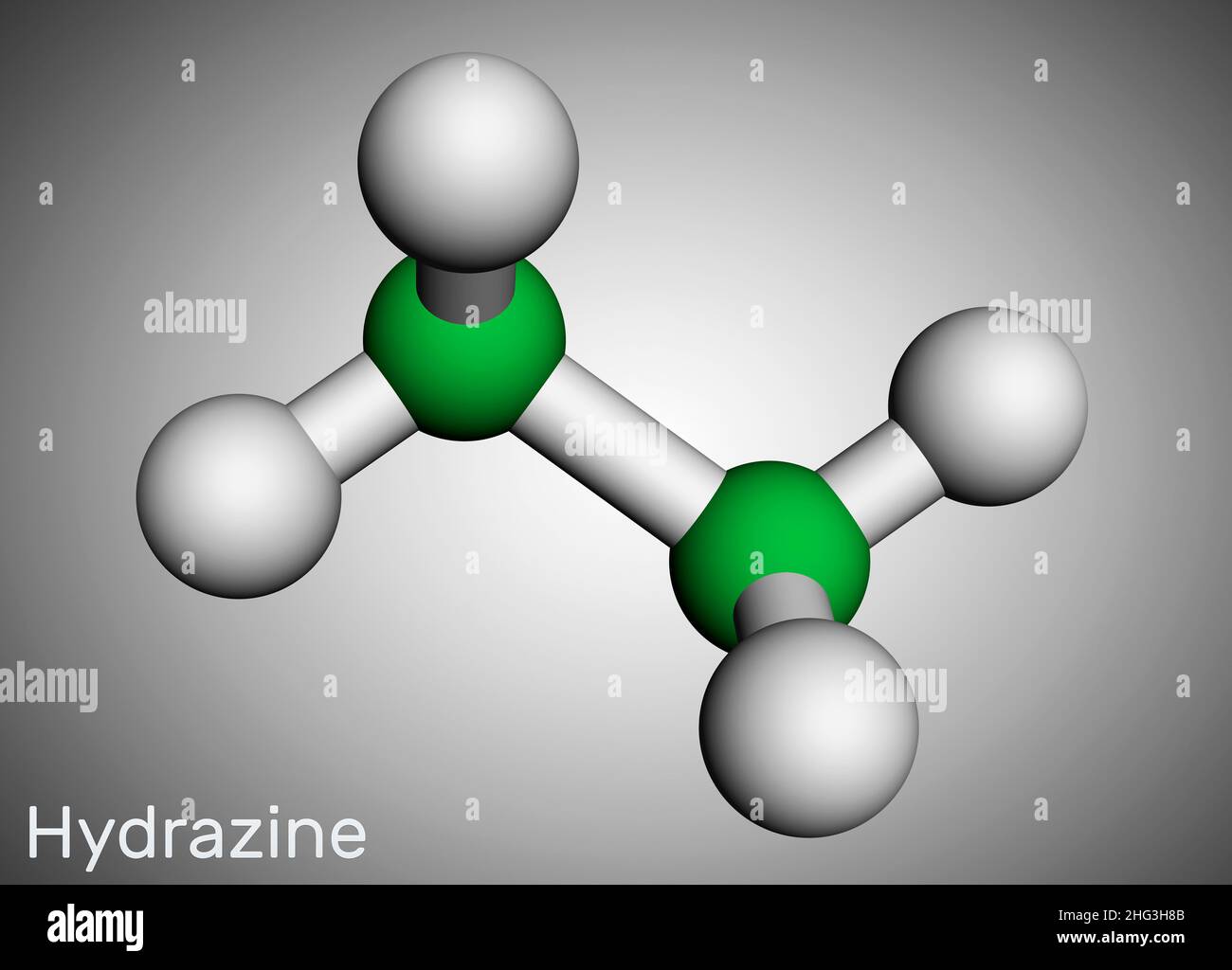
Hydrazine, diamine, diazane, N2H4 molecule. It is highly reactive base and reducing agent. Structural chemical formula and molecule model. Vector illu Stock Vector Image & Art - Alamy
Hydrazine, Diamine, Diazane, N2H4 Molecule. it is Highly Reactive Base and Reducing Agent. Molecular Model Stock Illustration - Illustration of nitrogen, fuel: 239580969
Hydrazine, diamine, diazane, N2H4 molecule. It is highly reactive base and reducing agent. Structural chemical formula and molecule model. Sheet of paper in a cage Stock Vector | Adobe Stock
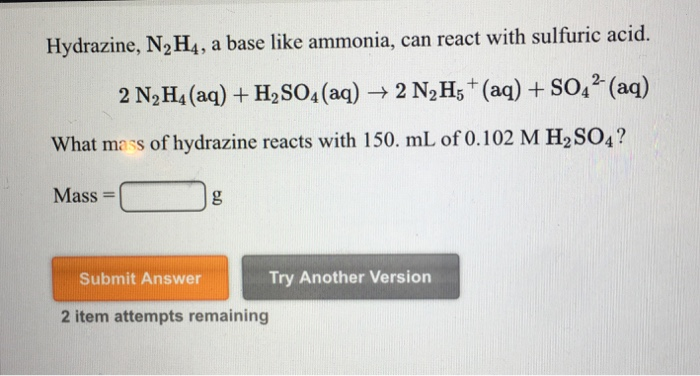
✓ Solved: Hydrazine, N2H4, is a weak base and can react with an acid such as sulfuric acid: 2N2H4(aq)+H2SO4(aq)→...

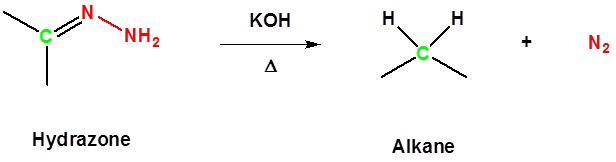




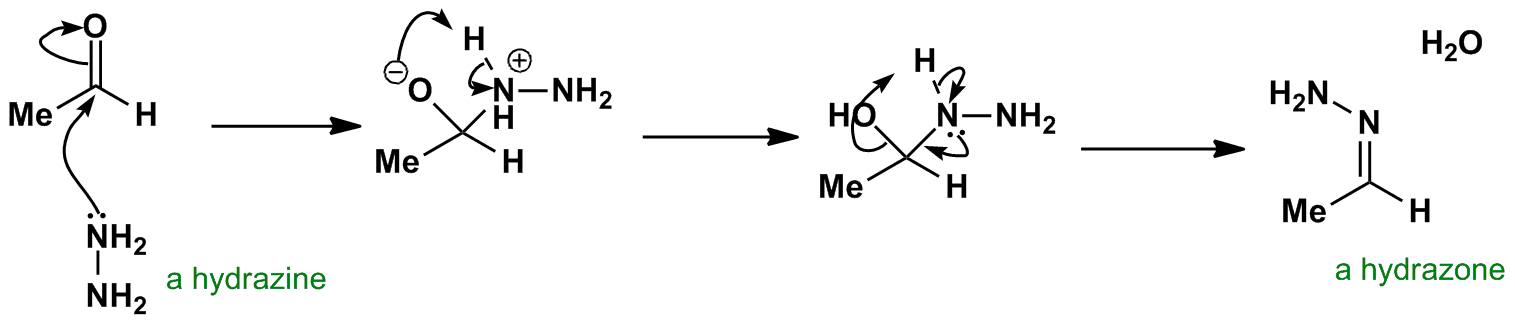



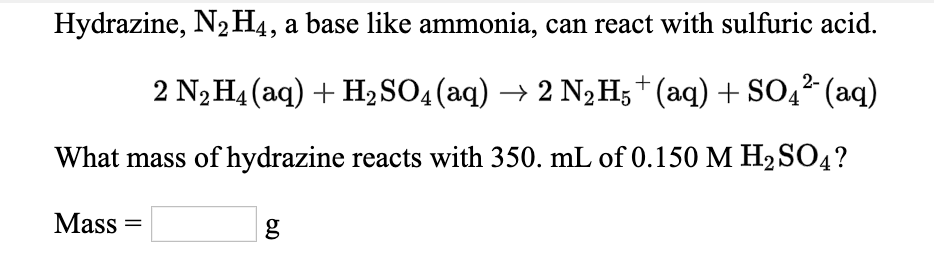
/chapter6/pages33and34/page33and34_files/WolffKishnermechanism.png)